Mr. Jeff Christopherson and Mr. Mike Roller demonstrated Halloween-themed experiments for their chemistry classes on Friday, Oct. 29, scaling the annual event back from previous years.
In his 5th-hour Honors Chemistry class, Christopherson exploded jack-o’-lanterns, lit students’ hands on fire, and popped 2-liter bottles decorated like Frankenstein’s monster.
Historically, the chemistry teachers combine classes to conduct the experiments and allow other students and staff members to observe the event. This year, the traditional Halloween festivities were performed just for the two teachers’ students.





























![Coach Drengwitz on the loss to Mt. Carmel, 2024 season [video]](https://nchsinkspot.com/wp-content/uploads/2024/11/Postseason-presser-feature-1200x800.png)

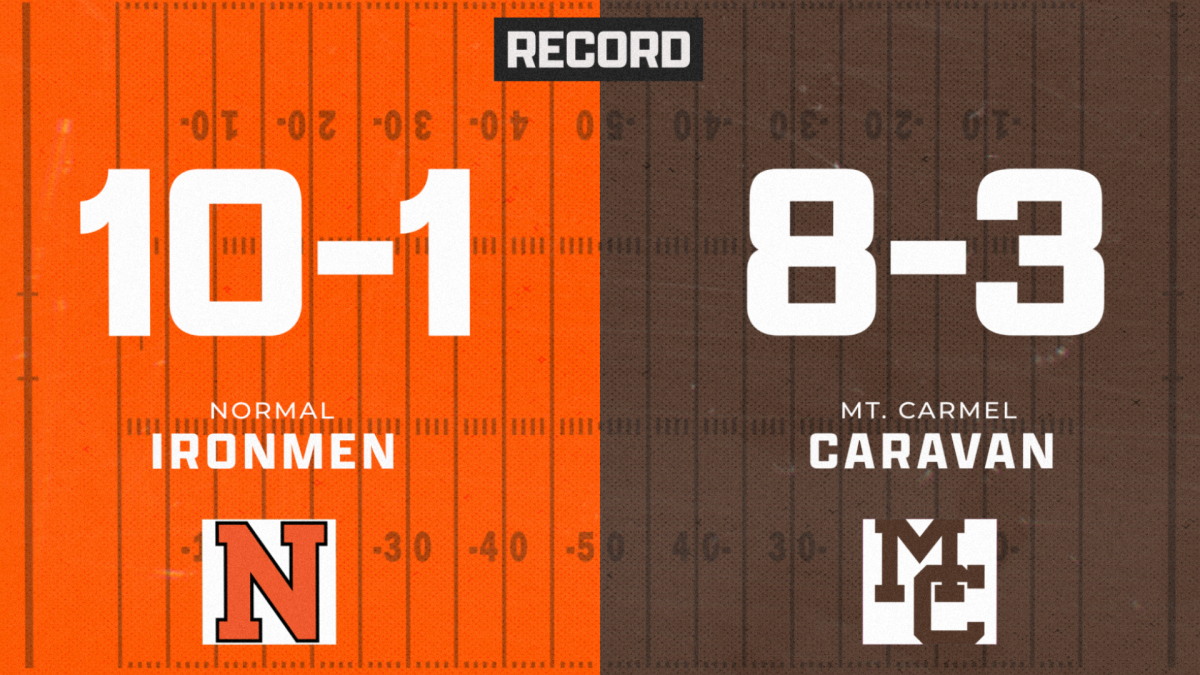
![IHSA 7A Football Playoffs Quarterfinals: Ironmen head coach on facing the Mt. Carmel Caravan [video]](https://nchsinkspot.com/wp-content/uploads/2024/11/0w12-web-feature-1200x800.png)










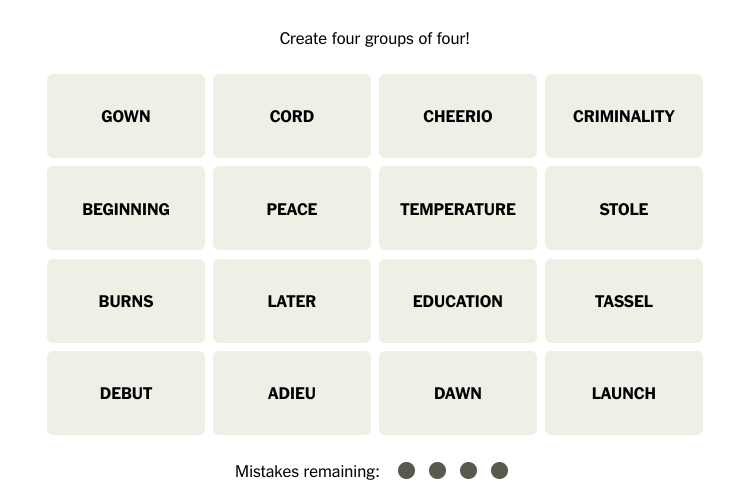
![Halloween candy cross section quiz [quiz]](https://nchsinkspot.com/wp-content/uploads/2022/10/Candy-cover-big-900x675.png)
![Average Jonah? [quiz]](https://nchsinkspot.com/wp-content/uploads/2022/05/average-jonah-900x600.png)


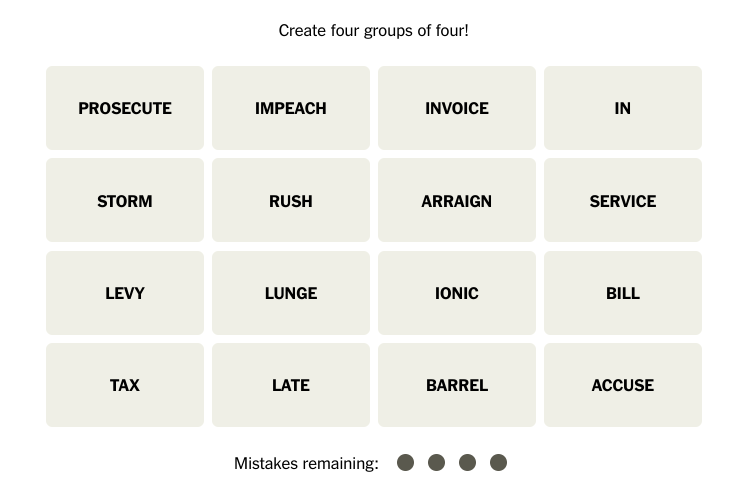
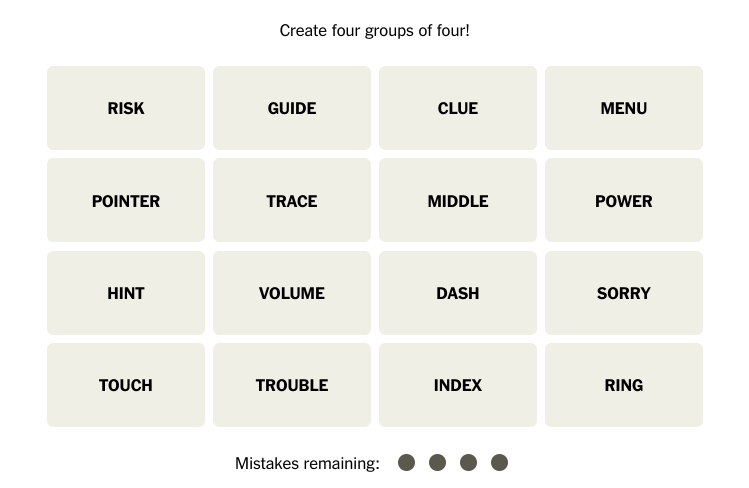















![Cell phone ban in schools? Community responds to proposed legislation [video]](https://nchsinkspot.com/wp-content/uploads/2025/04/Sequence.00_01_09_19.Still001-1200x675.png)




![Ironmen spring sports update: April 9 [video]](https://nchsinkspot.com/wp-content/uploads/2025/04/sports-recap-square-1200x1200.png)

![Ironmen in the hunt: Coach Feeney talks Big 12 Title race ahead of PND matchup [video]](https://nchsinkspot.com/wp-content/uploads/2025/01/feeney-1200x675.png)
![On the Spot: This or That – Halloween [video]](https://nchsinkspot.com/wp-content/uploads/2024/10/tot-Halloween-YT-1200x675.png)
![On the Spot: This or That – Fall favorites [video]](https://nchsinkspot.com/wp-content/uploads/2024/10/ots-fall-web-1200x800.png)
![On the Spot – Teachers tested on 2023’s hottest words [video]](https://nchsinkspot.com/wp-content/uploads/2024/01/On-the-Spot-Teachers-tested-1200x675.png)











